Chemistry of Seanol®

Molecular Structure of Sea Polyphenols
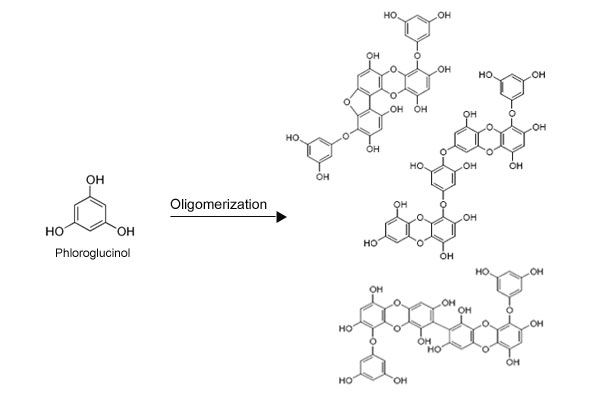
The uniqueness in chemistry of Sea polyphenols is that it is made of only one monomer called "phloroglucinol".
In other words, Sea Polyphenols are dehydro-oligomeric adducts of phloroglucinol.
Difference from Land-based Polyphenols
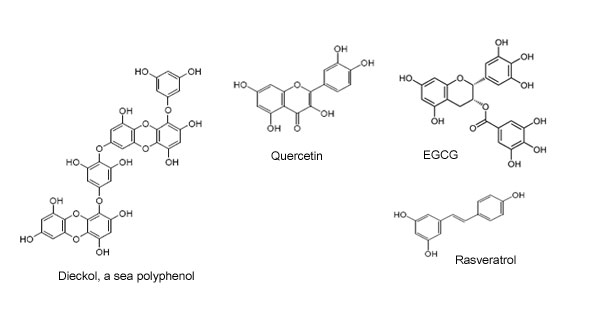
Multi-ring structure makes a Sea polyphenol molecule a highly potent electron trapping machine against any kind of free radical: Resveratrol (2 rings); flavonoids (3 rings), catechins (4 rings) Sea Polyphenols are chemically unique in that they do not have any other type of carbon than pure aromatic ones.
Biosynthesis
The exact whole biosynthetic pathway for a Sea polyphenol is unknown.
However, in the early biosynthetic steps of its monomer, phloroglucionol, two molecules of acetyl co-enzyme A are converted into malonyl co-enzyme A through the addition of carbon dioxide. This changes the acetyl methyl group into a highly reactive methylene and so assists the process of polymerization to occur without the need for a high investment of energy. During further synthesis steps, the carbon dioxide, which was added as an activator, is lost.
The result of polymerization is a polyketide chain consisting of an acid moiety, and the co-enzyme is lost. The polyketide chain is transformed by intermolecular ring closure and elimination of water to produce hexacyclic ring systems .
Triketide, the cyclization product, is not stable and thus undergoes transformation (tautomerisation) into the thermodynamically more stable aromatic form, phloroglucinol, consisting of three phenolic hydroxyl groups (Waterman PG and Mole S. 1994. Analysis of phenolic plant metabolites. Blackwell Scientific Publications: Oxford, Great Britain.
Further elements of the biosynthetic pathway are unknown.
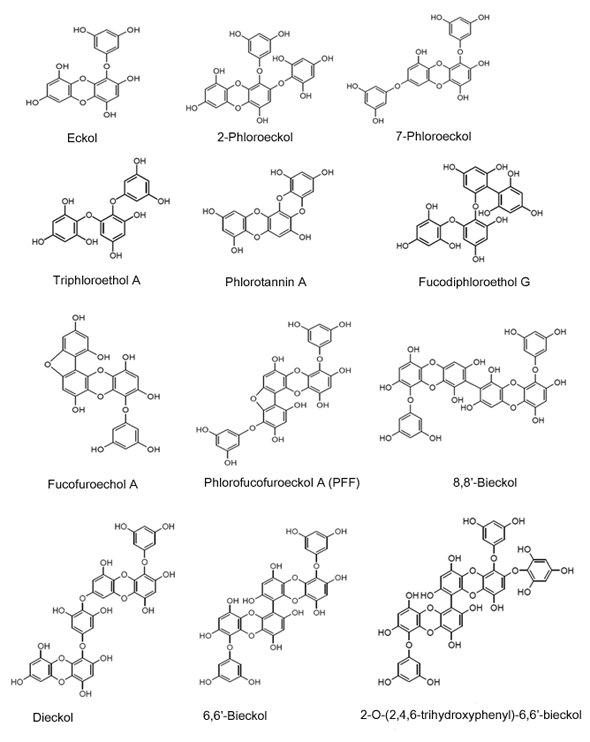
2D-Structures of Sea Polyphenols
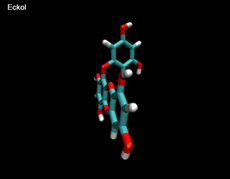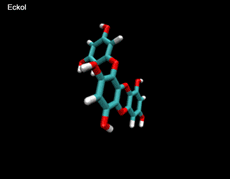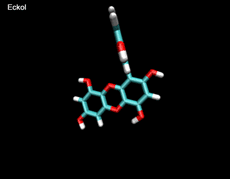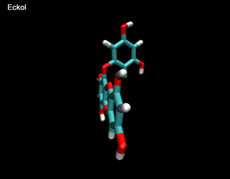
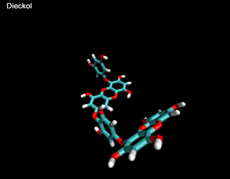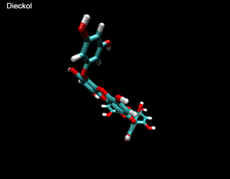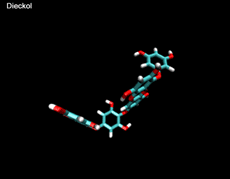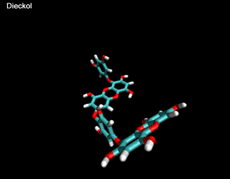
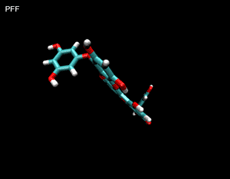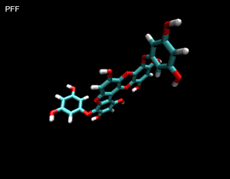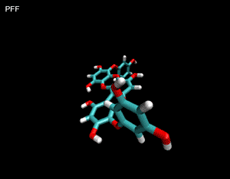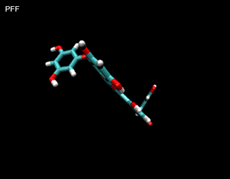
3D- Structures
3D-chemical structures of eckol, dieckol and phlorofucofuroeckolA have been calculated by using Density Functional Theory. Calculation were performed by Dr. J.G.Lee, Physics Department, North Carolina State University.